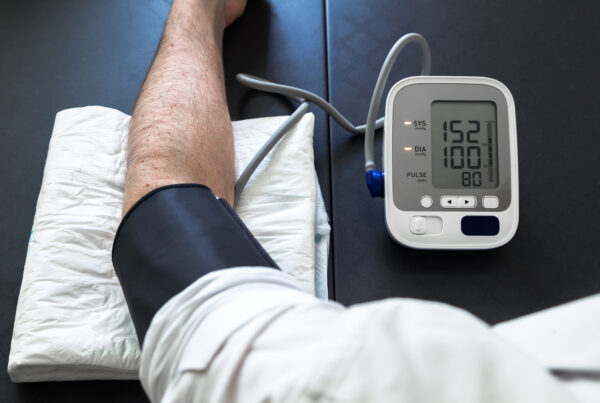
Blood pressure data done right starts with understanding the landscape; the regulatory requirements, the operational realities, and the standards that matter when it counts.
This is where we share what we know. Browse our latest thinking, published research, case studies from live studies, and practical guides to help clinical teams make informed decisions about BP data collection and management in trials.
Blood Pressure Monitoring
We support a full range of blood pressure monitoring methods
to suit diverse study designs and patient populations:
Ambulatory Blood Pressure (ABPM)
24-hour monitoring with validated devices and built-in compliance tracking.
Home Blood Pressure (HBPM)
Enables remote, patient-driven data collection with minimal burden.
Office Blood Pressure (OBP)
Also known as Clinic Blood Pressure, these are standardised clinical-site readings with real-time data uploads.
Central Blood Pressure (CBP)
CBP measures the blood pressure in the aorta, the large artery directly connected to the heart.
Guidelines
ABPM
What is ABPM?
Ambulatory Blood Pressure Monitoring (ABPM) measures blood pressure (BP) over 24 hours, during daytime and night-time periods, while patients carry out their normal activities and sleep. It provides multiple readings as the patient goes about their day, offering a more accurate picture of BP behaviour compared to office measurements alone.
Why use ABPM in Clinical Trials?
The European Hypertension Guidelines recommend ABPM as a crucial tool for accurate BP diagnosis, management, and risk assessment. It helps identify white-coat and masked hypertension, monitors treatment effectiveness and improves it cardiovascular risk prediction more effectively than office BP alone.
dabl Clinical Trials provides 24-hour Ambulatory Blood Pressure Monitoring (ABPM). This allows you to collect real-time, precise ABPM data using regulated, dependable devices and centralised data analysis.
Home Blood Pressure (Home BP)
What is Home Blood Pressure?
Home Blood Pressure (Home BP) refers to the regular monitoring and recording of blood pressure by patients themselves. This is typically done by using an automated blood pressure device in a non-clinical setting, usually at home.
The European Hypertension Guidelines define Home BP as an important method for assessing out-of-office blood pressure, alongside ABPM. This method provides valuable insights into a patient’s blood pressure trends outside clinic visits, offering an accurate reflection of everyday cardiovascular health.
Home BP monitoring:
- Improves diagnostic accuracy
- Better predicts cardiovascular outcomes than office BP
- Empowers patients in self-care and adherence
- Useful for long-term monitoring
Why use Home BP in Clinical Trials?
The European Hypertension Guidelines recognise Home BP Monitoring as a simple, cost-effective and clinically valuable method for diagnosing and managing hypertension. It is particularly useful for detecting white-coat and masked hypertension and for ongoing treatment monitoring.
dabl Clinical Trials provides Home BP monitoring to capture real-time validated data. We provided bespoke training and support. The accurate, dependable data we collect helps your trial to evaluate effectiveness of interventions, to identify adverse events and to measure primary or secondary endpoints, such as average systolic/diastolic BP over time. It’s convenient for the patient and provides a rich dataset for analysis.
Office Blood Pressure
What is Office Blood Pressure?
Office blood pressure (Office BP) is blood pressure measurement taken in a clinical setting by a healthcare professional, usually in a doctor’s office, in a hospital or in a clinic.
The European Hypertension Guidelines recommend proper standardised technique for Office BP and to confirm diagnosis of hypertension with ABPM or HBPM where possible.
dabl Clinical Trials provides Office BP via a validated, centralised and standardised platform. This ensures accuracy, consistency and regulatory compliance when it comes to measuring and reporting on blood pressure data across multiple sites
Recent Blog Articles

White Coat Hypertension: Diagnosis, Risks and the Role of ABPM

Where We've Been
Publications Supported by dabl
The evidence base for rigorous BP measurement in clinical trials spans decades of research, landmark outcomes studies, and evolving regulatory guidance.
Browse our curated collection of published literature from trials where blood pressure has featured as a primary or secondary endpoint; from large-scale cardiovascular outcomes studies to device validation research.
A useful reference point for clinical teams designing studies, selecting endpoints, or building the case for standardised BP data collection.